Hepatotoxicity Assay Services
Our cytotoxicity and hepatotoxicity screening service makes early, high-acuity assessment of hepatotoxic risk cost-effective for smaller organizations and thereby brings new illumination to the challenge of reducing late-stage candidate attrition.
Background
- Hepatotoxicity is a critical concern in drug development because the liver is the primary organ responsible for drug metabolism and detoxification.
- In vitro hepatocyte models are widely used to assess hepatotoxicity during drug discovery and development.
- Primary hepatocytes retain key liver-specific functions, including drug-metabolizing enzymes, transporters, and metabolic pathways, making them a physiologically relevant system for evaluating compound-induced cytotoxicity, mitochondrial dysfunction, and metabolic liabilities.
- The HUREL® micro liver models (primary hepatocytes co-culture with stromal cells) are effective for screening acute and chronic hepatotoxicity, as they maintain stable metabolic functions for over four weeks.
- These models enable early identification of hepatotoxic risk, support mechanistic investigations, and help guide compound optimization while reducing reliance on animal studies.
General Procedure
- HUREL® micro liver models are prepared by co-culturing primary hepatocytes and stromal cells and cultured for one week prior to dosing compounds.
- Treatment with test compounds
- Hepatotoxicity is assessed using an ATP cell viability assay after 7-day or 14-day exposure to the test compounds.
- TC₅₀ values are calculated from non-linear regression curves fitted with GraphPad software.
- Hepatotoxicity is calculated using the DILI index [1,2].

Protocol
| Cell Model | HUREL® Primary Hepatocyte Micro Liver Models (Species: Human, Monkey, Dog, Rat, Mouse, Minipig, Rabbit, etc) |
| Time Points | t = days 7 or 14 after dosing (custom time points available |
| Test Article Concentration | 6 different concentrations, including vehicle control (custom concentrations available) |
| Positive Controls | Cyclophosphamide (custom controls available) |
| Number of Replicates | 3 replicates per time condition |
| Assay Method | ATP cell viability assay (Cell-Titer Glo from Promega) (custom assays available: ALT, AST, LDH, etc) |
| Analysis Method | Luminescence measurement using microplate reader |
| Test Article Requirements | 50 µL of ~50 mM solution or equivalent amount of solid |
| Data Delivery | TC50 values, heat map, raw data, report |
Data
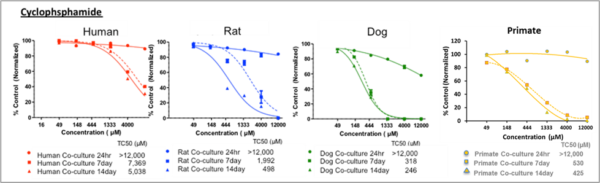
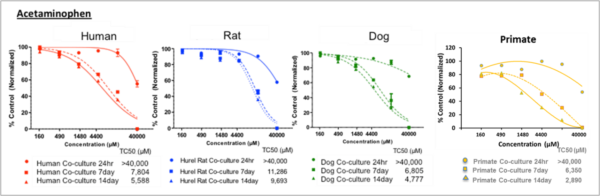
Figure 1. Dose-dependent cytotoxicity after single and repeated treatments in HUREL primary hepatic co-cultures. Non-linear regression curves estimating dose-dependent cytotoxicity after single and repeated treatments of the HUREL human, monkey, dog, and rat hepatic co-cultures with acetaminophen and cyclophosphamide, respectively. In each instance the greatest increase in cytotoxicity was measured between 24 h and Day 7, with some additional increase measured between Day 7 and Day 13. Data from Novik et al (2017), Toxicology and Applied Pharmacology [3].
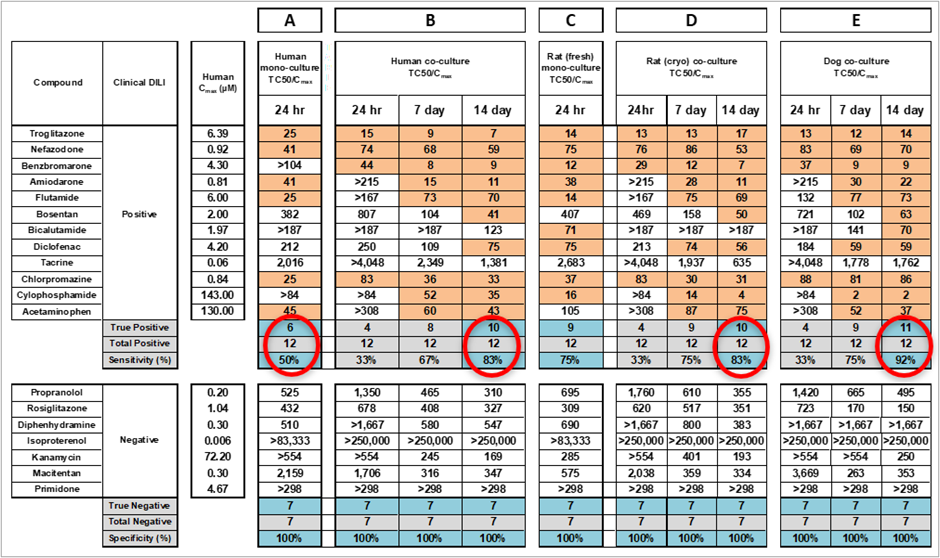
Table 1. Sensitivity and specificity of cytotoxic response to compounds of Sample Set I. A: Mono-culture of human cryopreserved primary hepatocytes after 24-hour single-dose treatment. B: HUREL co-culture of human cryopreserved primary hepatocytes after 24-hour single-dose treatment and at Days 7 and 13 after repeat-dose treatment every 48 h. C: Mono-culture of rat fresh primary hepatocytes after 24-hour single-dose treatment. D: HUREL co-culture of rat cryopreserved primary hepatocytes after 24-hour single-dose treatment and at Days 7 and 13 after repeatdose treatment every 48 h. E: HUREL co-culture of dog cryopreserved primary hepatocytes after 24-hour single-dose treatment and at Days 7 and 13 after repeat-dose treatment every 48 h. Data from Novik et al (2017), Toxicology and Applied Pharmacology [3].
References
- O’Brien, P.J., Irwin, W., Diaz, D. et al. High concordance of drug-induced human hepatotoxicity with in vitro cytotoxicity measured in a novel cell-based model using high content screening. Arch Toxicol 80, 580–604 (2006).
- Jinghai J. Xu, Peter V. Henstock, Margaret C. Dunn, Arthur R. Smith, Jeffrey R. Chabot, David de Graaf, Cellular Imaging Predictions of Clinical Drug-Induced Liver Injury, Toxicological Sciences, Volume 105, Issue 1, September 2008, Pages 97–105
- Novik E, Dwyer J, Morelli J, Parekh A, Cho C, Pludwinski E, Shrirao A, Freedman R, MacDonald J and Jayyosi Z. “Long-enduring primary hepatocyte-based co-cultures improve prediction of hepatotoxicity”. Toxicology and Applied Pharmacology, 336 (2017) 20-30

